- Part 1: The Building Blocks – Understanding the Electron’s Energy
- Part 2: The Electron in Action – Flow, Punch, and Color
- Part 3: The Grand Finale – Forging an X-Ray
- Inside the X-Ray Tube: A Controlled Collision
- Conclusion
The electron. It’s the unsung hero of our modern world, powering everything from your phone to the lights in your home. We talk about electricity in terms of Volts and Amps, but what do those words really mean for a single electron? What gives one electron a gentle nudge versus a punch powerful enough to create a medical X-ray?
Join us on a journey as we follow a single electron, starting with the basics of its energy and ending with the violent collision that allows us to see inside the human body.
Part 1: The Building Blocks – Understanding the Electron’s Energy
Before we can make an X-ray, we need to understand the language of electron energy. Think of electron volts (eV) as the “energy” or “punch” of a single electron, while current (Amps) is how many electrons are moving at once.
Two electrons can have the same energy (e.g., 10 eV), but if one is moving through a vacuum and another through a material where it interacts with atoms, their speeds can differ due to collisions and resistance.
This shows that electron volt (eV) measures energy (the “punch”), not the actual speed of the electron.
What is an Electron Volt (eV)?
In the simplest terms, an electron volt is the amount of energy one single electron gains when it passes through a 1-volt battery.
1 eV is the energy an electron gains when accelerated through a potential difference of 1 volt.
If you have a standard 9V battery, Ideally, every single electron that flows out of it has been given a 9 eV kick of energy to spend on whatever it’s powering.
Think of it like a water slide:
- Voltage (Volts) is the height of the slide. A 12-volt slide is much higher than a 1.5-volt slide.
- Electron Volt (eV) is the energy one person (the electron) has when they hit the water at the bottom. The higher the slide, the bigger the splash.
- The key difference is that Volts are a property of the source (the battery or wall outlet), telling you how much “push” is available. The eV is the property of the particle, telling you how much energy one electron actually “bags” from that push.
Why Do Scientists Use eV? A Matter of Scale
In our daily lives, we measure energy in Joules (like the calories in food). But for a single electron, a Joule is a ridiculously huge unit—it’s like trying to measure the weight of a single grain of sand in tons.
To give you an idea, 1 Joule is roughly 6.24 quintillion electron volts (6.24 x 10¹⁸ eV).
Scientists created the eV to use small, manageable numbers. It makes the math simple: an electron moving through a 100-volt circuit gains 100 eV of energy. Done.
Part 2: The Electron in Action – Flow, Punch, and Color
So, we know how to measure an electron’s energy. But what can different amounts of energy actually do? This is where we bring in its partner: Current (Amps), which is simply the “flow” or number of electrons moving at once.
| Component | Common Voltage (The “Push”) | Common Current (The “Flow”) |
|---|---|---|
| AA Battery | 1.5 Volts | ~50–500 mA (Small flashlight) |
| USB Charger | 5 Volts | 1–3 Amps (Phone charging) |
| Wall Outlet | 230 Volts | 5–15 Amps (Toaster/Kettle) |
| Car Battery | 12 Volts | 50–400 Amps (Starting the engine) |
The big difference is this: to light a blue LED, an electron needs about 3 eV of “punch.” A single 1.5V AA battery isn’t enough to give it that kick. But to make the LED bright enough to see, you need millions of billions of these electrons flowing every second (the current).
Voltage (eV) determines the type of work done (e.g., the color of light), while Current (Amps) determines the intensity (the brightness).
Here’s a rough guide to the electron’s power level:
| Energy | What it can do (Layman’s Terms) | Real-World Example |
|---|---|---|
| ~1 eV | The “Nudge”: Enough to move through a copper wire or create infrared (heat) light. It can’t make visible light yet. | The warmth from a radiator or the invisible beam from a TV remote. |
| 2–3 eV | The “Glow”: The sweet spot for human vision. This energy is needed to “kick” a photon of visible light into existence. | Red light needs ~1.8 eV; Blue light needs ~3.1 eV. |
| >10 eV | The “Punch”: Enough energy to rip an electron away from its atom (ionization). This creates Ultraviolet (UV) light. | Energy strong enough to cause sunburns or other chemical changes. |
Part 3: The Grand Finale – Forging an X-Ray
Now, what happens when we take that “punch” and turn it into a bone-shattering uppercut? We create an X-ray.
High Energy = High Frequency
A common mistake is thinking “stronger” means “bigger.” In the world of particles and waves, higher energy equals higher frequency (a shorter, faster wave).
Imagine you are shaking a long rope:
- Low Energy: If you move your hand slowly, you create lazy, long waves (Low Frequency). This is like a radio wave.
- High Energy: If you shake your hand violently and rapidly, you create tight, short, frantic waves (High Frequency). This is like an X-ray.
- When we give an electron an enormous amount of energy—say, 100,000 eV (from a 100 kV power source)—and slam it into a target, its rapid deceleration near atomic nuclei converts its energy into a high-frequency photon: an X-ray.
Inside the X-Ray Tube: A Controlled Collision
To create X-rays, we fire a high-current beam of high-energy electrons at a metal target inside a vacuum tube. But the target can’t be just any metal. It has to be Tungsten.
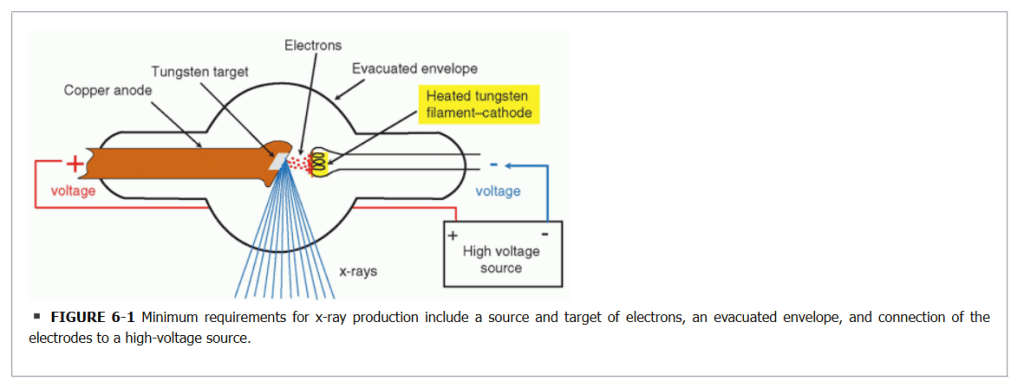
Why Tungsten?
For two critical reasons:
- High Atomic Number (Z=74): Tungsten’s nucleus is massive, packed with 74 protons. This creates a powerful positive charge that is extremely effective at grabbing and “braking” the incoming high-speed electrons, forcing them to shed their energy as X-rays.
- Insanely High Melting Point (3,422°C): This process is incredibly inefficient. About 99% of the electron’s energy is lost as pure heat. Any lesser metal, like aluminum (melting point 660°C), would instantly turn into a puddle. Tungsten can take the abuse and stay solid while glowing white-hot.
The Two Ways to Make an X-Ray Photon
When a 100,000 eV electron hits the Tungsten target, its energy is released in one of two ways. This is a game of quantum probability.
- The “Near-Miss” (Bremsstrahlung or “Braking Radiation”): This is the most common outcome. The incoming electron doesn’t hit anything directly but flies close to a Tungsten nucleus. The nucleus’s massive positive charge “tugs” on the negative electron, causing it to swerve and slow down—or “brake.” The kinetic energy it loses is instantly converted into an X-ray photon. This process is messy, creating a wide spectrum of X-ray energies and accounting for that 99% heat loss.
- The “Direct Hit” (Characteristic X-Rays): This is rarer but more precise. The incoming electron has so much energy that it slams directly into an electron in Tungsten’s inner orbit (the “K-shell”) and knocks it out of the atom.
- The Gap: This leaves a hole in a low-energy orbit, which nature hates.
- The Jump: An electron from a higher, outer orbit immediately “falls” down to fill the gap.
- The Photon: To fall into a lower-energy shell, the electron must shed the energy difference. It does so by spitting out a photon of a very specific, predictable energy (or frequency). This is a “characteristic” X-ray, unique to the Tungsten atom.
Conclusion
From a single electron volt gained in a battery to the 100,000 eV punch needed to forge an X-ray, the journey of the electron is one of escalating energy and impact. By understanding how to control its “push” (Voltage) and its “flow” (Amps), we’ve harnessed this tiny particle’s power—turning the rapid deceleration of high-energy electrons into X-rays that allow us to see inside the human body.
Leave a comment