Table of Content
- Is Electron a Real Cloud of Charge or a Ghost of Probability?
- The “Electron Cloud”: A Powerful Model for Chemistry
- The Problem with a “Real” Cloud: The Physicist’s Objection
- The “Probability Wave”: The Fundamental Reality
- How two Atoms bond through electrons
- From Fuzzy Wave to Single Point: The Collapse
- Reconciling the Views: Why the “Wrong” Model is So Right
- How This “Effective Cloud” Creates Molecules
- The Reality of “Empty Space”
- Conclusion
Is Electron a Real Cloud of Charge or a Ghost of Probability?
When we try to picture an electron, the fundamental particle that powers our world, we often hear two conflicting descriptions: is it a fuzzy “cloud of negative charge” or a mathematical “wave of probability”? This isn’t just a matter of semantics; it touches on a deep divide between how we model the world and what might be its fundamental reality.
The short answer is that the “cloud” is an incredibly powerful and predictive model, especially in chemistry, while the “probability wave” is a more technically accurate description of the underlying quantum physics. Understanding the difference—and why the “wrong” model works so well—is key to decoding the true nature of matter.
In quantum mechanics, an electron isn’t a tiny billiard ball orbiting a nucleus. Its state is described by a mathematical function called a wavefunction (denoted by the Greek letter ψ). This function is the key to understanding its strange dual identity.
The “Electron Cloud”: A Powerful Model for Chemistry
The “cloud” model is a visual representation of where an electron is likely to be. If you were to take a trillion “snapshots” of an electron’s position in an atom and overlay them, you would see a fuzzy, cloud-like shape.
- Dense areas: High probability of finding the electron.
- Thin/Transparent areas: Low probability of finding the electron.
This “cloud” is an intuitive way to think about orbitals. For example, in a simple Hydrogen atom (the 1s orbital), the “cloud” is a perfect sphere. In more complex atoms, these clouds take on shapes like dumbbells or donuts.
This model is the bedrock of modern chemistry because it perfectly predicts how atoms will behave and bond.
The Problem with a “Real” Cloud: The Physicist’s Objection
Here’s the catch: it is a common mental model to think of the electron as a physical “cloud of charge” (like a mist or a fluid), but experiments show that this isn’t quite what’s happening. If the electron were a real, physical cloud of “electron-stuff,” we would run into two major problems:
- The “Piece” Problem: When you measure an electron, you would expect to find a piece of the cloud or a fraction of its charge. But this never happens. We always find the entire electron—full charge, full mass—in one single, specific spot. This repeatable, verified experimental fact proves that the electron is not a physical mist that can be divided.
- The “Repulsion” Problem: If the electron were a physical cloud of negative charge, one part of the cloud would repel the other parts. The cloud should instantly blow itself apart. It doesn’t.
This tells us that while the “cloud” shape is a fantastic predictor of behavior, it isn’t a literal description of the electron’s substance.
The “Probability Wave”: The Fundamental Reality
Instead of a cloud of matter, physicists describe the electron using its wavefunction, which is a “probability amplitude cloud.” This is a crucial distinction.
- Unobserved State (Superposition): The electron isn’t “smeared out” like a physical fluid. Instead, it exists in a superposition of possibilities. It has the potential to be at Point A, Point B, and Point C all at once. It exists in a superposition of possible states, which allows it to behave as if it were in multiple locations when measured statistically. A physical cloud of gas is in one state (the “gas state”).
- The Math of Probability: We use the wavefunction (ψ) to calculate the probability of finding the electron in a specific region. This is governed by the Born Rule, where the probability density is given by the square of the absolute value of the wavefunction: P = |ψ|². The “cloud” is just a 3D graph of this probability.
Because it is a superposition of waves, it can do things a fluid can’t—like interfere with itself. This wavefunction behaves like a wave mathematically and can exhibit interference, though whether it represents a physically real entity or just information is still debated.
How two Atoms bond through electrons
- When two atoms approach each other, their electrons are no longer confined to separate regions—they are described by wavefunctions that can overlap and combine.
- According to Molecular Orbital Theory, these wavefunctions form new orbitals: a bonding combination (ψ₁ + ψ₂) and an antibonding one (ψ₁ − ψ₂). In the bonding case, the electron probability becomes concentrated between the two nuclei.
- This doesn’t mean a physical cloud is merging, but that the probability of finding the electrons is highest in the region between the nuclei, allowing them to interact with both atoms at once.
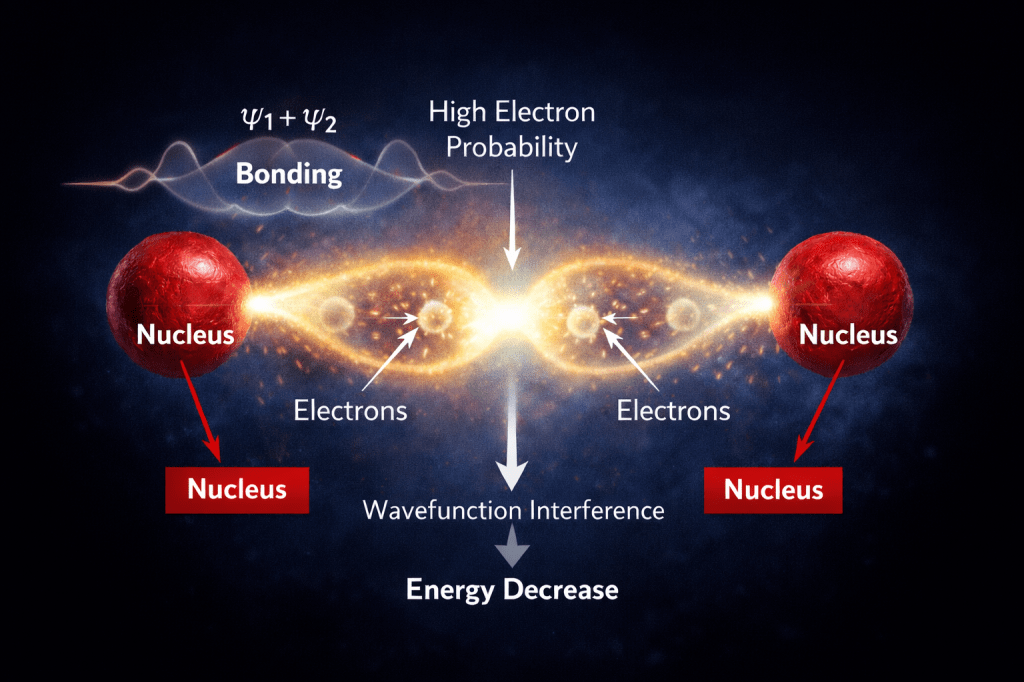
This redistribution of probability lowers the system’s energy.
- First, the electrons in between the nuclei experience stronger attraction to both positively charged protons simultaneously, which reduces potential energy.
- Second, the combined wavefunction becomes smoother in that region, lowering its curvature and therefore its kinetic energy (as described by the Schrödinger equation).
- These two effects together outweigh the natural repulsion between the nuclei, creating a net energy decrease.
A bond forms because physical systems tend toward lower energy states.
- When two electrons occupy this lower-energy bonding orbital (with opposite spins), they stabilize the arrangement, effectively “holding” the nuclei together.
- If instead the electrons occupied the antibonding orbital, where probability is pushed away from the space between nuclei, the energy would increase and no bond would form.
- Thus, bonding is fundamentally the result of wavefunction interference that concentrates probability between nuclei and lowers total energy.
From Fuzzy Wave to Single Point: The Collapse
The “wave” or “cloud” phase ends the moment you interact with the electron to “see” where it is (a measurement).
This is known as the collapse of the wavefunction.
- The Transition: The broad, fuzzy wave of possibilities “snaps” into a single, localized point. You aren’t “compressing a cloud”; you are collapsing the math. In the standard interpretation, measurement causes the wavefunction to collapse into a definite outcome.
- The Location: Where it appears is random, but the density of the probability wave tells you the likelihood. If the “cloud” is thick in the center, you are much more likely to find the “point” there.
- The Catch: After measurement, the wavefunction evolves again over time according to the Schrödinger equation, typically spreading out from its localized state.
A helpful analogy is the “haze” of a guitar string. Imagine a string vibrating so fast it looks like a blurry “haze.” While unobserved, the vibration occupies a whole area. This “haze” is its reality. If you suddenly pinch the string, you have “localized” it to a single point under your finger.
| State | Behavior | What It Really Is |
|---|---|---|
| Unobserved | Distributed Wave | A fuzzy cloud of probability existing in a superposition of states. |
| Observed | Localized Particle | A specific point-like coordinate in space after the wavefunction collapses. |
The transition from a “cloud of potential” to a “point of reality” is one of the biggest mysteries in physics, often called the Measurement Problem.
Reconciling the Views: Why the “Wrong” Model is So Right
So, which is true? Fundamentally, the electron is a quantum object described by a mathematical wavefunction (the “probability wave”).
But if the physical cloud idea is wrong, why is it so common and powerful in fields like chemistry?
Because even though the “physical cloud” idea is fundamentally incorrect, it is operationally and predictively right for a huge range of phenomena.
The average effect of the probability wave over time is indistinguishable from a cloud of negative charge with a specific shape.
- Atomic Repulsion: When you push on a table, the electron probability waves in your hand get close to the probability waves in the table. The math that governs how these waves interact results in a repulsive force that is exactly what you would calculate if you treated them as two physical clouds of negative charge pushing against each other.
- Chemical Bonding: When two hydrogen atoms form a molecule, their individual wavefunctions merge. The new, combined probability is highest between the two nuclei. This region of high probability creates a net attractive force holding the atoms together, acting precisely like an “electron glue” or a merged “charge cloud.”
The best way to reconcile the two is this:
The electron is fundamentally a quantum object described by a wavefunction (the physicist’s probability wave).
When measured, it collapses to a point.
However, when left alone in an atom, the physical consequences of its wavelike existence are perfectly described by treating it as an effective cloud of charge (the chemist’s practical model).
One is the fundamental reality. The other is the emergent physical behaviour.
How This “Effective Cloud” Creates Molecules
Now that we understand why the cloud model works, we can see how it determines how atoms bond to form molecules. When atoms come together, their individual electron “clouds” (orbitals) merge and reshape themselves in a process called Orbital Hybridization.
- Think of it like two soap bubbles touching: they don’t stay as two separate spheres; they fuse into a new, single shape.
- The Merge (Covalent Bonding): When two atoms get close, their electron clouds overlap. The two separate “probability waves” combine into one larger cloud that encompasses both nuclei. This shared cloud is a bonding orbital, and its density between the two nuclei acts like a “glue” holding them together.
- Reshaping the Cloud (Hybridization): Carbon is the master of this. It often mixes its separate spherical (s) and dumbbell-shaped (p) orbitals to create four identical “sp3” hybrid clouds. These new clouds point towards the corners of a tetrahedron, which is why a diamond or a methane molecule has the specific 3D shape it does.
The specific shape of these merged clouds determines almost everything about the world around us:
| Shape | Example | Result |
|---|---|---|
| Bent | Water (H2O) | The “V” shape makes water polar, allowing it to dissolve salt and support life. |
| Linear | Carbon Dioxide (CO2) | The straight line makes it non-polar and a gas at room temperature. |
| Planar | Graphite | Flat sheets of clouds allow layers to slide over each other, making it a good lubricant. |
The Reality of “Empty Space”
- Even though we say an atom is 99.9% empty space, it doesn’t feel empty precisely because of these clouds. When you touch a table, you aren’t feeling “solid” matter.
- You are feeling the electrostatic repulsion of the effective electron clouds in your hand pushing against the effective electron clouds in the wood.
- You are, in essence, hovering on a cushion of quantum probability. The ghost of possibility has created the solid world.
Conclusion
An electron is not a tiny solid particle nor a literal cloud of charge—it is fundamentally described by a wavefunction, a mathematical object that encodes the probability of where it might be found.
The familiar “electron cloud” used in chemistry is a highly effective visualization of this probability distribution, showing where an electron is likely to exist if measured many times. While this cloud model is not a physically real mist of charge, it works remarkably well because the average behavior of the electron’s wavefunction produces effects that closely resemble a spread-out charge distribution, allowing us to predict atomic structure, bonding, and material properties with great accuracy.
- When atoms come together, their electron wavefunctions overlap and reorganize into new patterns, concentrating probability between nuclei in bonding states and lowering the system’s energy.
- This redistribution acts like a stabilizing “glue,” holding atoms together to form molecules and ultimately shaping the physical world around us.
Takeaway: the solid, tangible reality we experience—chemical bonds, materials, and structure—emerges from an underlying quantum world governed not by definite positions, but by probabilities, wave-like behavior, and energy minimization.
Leave a comment