Table of Content
- Have you ever wondered what really holds the world together?
- Part 1: The Atom’s Hotel – Rule 1: No Two Tenants are Alike
- Pauli Exclusion Principle
- Part 2: The Shape of the Wave – What a “Room” Really Is
- Part 3: The Cosmic Handshake – Rule 2: Waves Love to Harmonize
- Constructive Interference (The “Glue”)
- Destructive Interference (The “Wall”)
- Part 4: The Rules in Action – Who Gets to Bond?
- Hund’s Rule
- Part 1: The Atom’s Hotel – Rule 1: No Two Tenants are Alike
- Case Studies
- The Case of Nitrogen (Atomic Number 7)
- The Case of Oxygen (Atomic Number 8)
- The Case of Helium (2) and Neon (10)
- The Curious Case of Carbon (Number 6) : The quirky Behavior
- The Starting Point (Ground State)
- The Carbon Twist (Excitation & Hybridization)
- The Result: Four “Lonely” Guests
- Why Carbon “Promotes” Electrons (and why others don’t always)
- Why not others?
- 5. Silver Atoms (Atomic Number 47) and Hund’s Rule
- The Big Picture
Have you ever wondered what really holds the world together?
We’re told that atoms are the building blocks of everything, but that just pushes the question back.
- What rules govern the atoms themselves?
- Why is silver chemically reactive, but helium isn’t?
- Why is a water molecule bent into a “V” shape, and not a straight line?
- And how does carbon, a simple element, manage to build the impossibly complex chains of DNA?
- The answer isn’t a long list of complicated chemical laws.
- In fact, most of reality can be explained by two bizarre, yet beautifully simple, quantum rules.
If you’ve ever felt that science is just a collection of facts to memorize, this article is for you. We are going on a journey to the heart of matter. By the end, you won’t just know that things happen; you will understand why they happen, using the fundamental logic of the universe itself.
Part 1: The Atom’s Hotel – Rule 1: No Two Tenants are Alike
Let’s start with a common point of confusion: we learn an atomic orbital (a “room”) can only hold two electrons. So how does an atom like Silver hold 47?
The answer is to think of an atom not as a single room, but as a giant, multi-story hotel. The electrons are the tenants, and they fill the hotel according to one strict rule, known as the Pauli Exclusion Principle.
Pauli Exclusion Principle
No two electrons in an atom can have the exact same “address.” They must differ in some way.
The Pauli Exclusion Principle is a fundamental rule of nature that prevents two electrons from being identical in every way.
Think of every electron as having a specific quantum state or “address” based on its energy level, its orbital type (1s, 2s, 2px, etc.), and its direction of spin.
The Rule
No two electrons in the same atom can occupy the exact same state at the same time.
How it Works in Orbitals:
- The Capacity: Each individual orbital (like 2px) has room for exactly two electrons.
- The Difference: To share that same orbital, the two electrons must have opposite spins. One must be “Spin Up” and the other must be “Spin Down”.
- The Limit: If a third electron tries to enter that same 2px orbital, it would be forced to have a spin that matches one of the two already there. Since they cannot have the same “address” (same orbital + same spin), the third electron is excluded and must move to a different, higher-energy orbital.
The Bottom Line: This principle is why electrons stack up into different shells and orbits rather than all crowding into the center. It creates the structure of the entire periodic table.
The Hotel Layout (Shells and Orbitals)
Like a hotel, an atom has floors (called Shells) and different types of rooms (called Subshells or Orbitals). As you move up the floors (further from the nucleus), the floors get bigger and can hold more rooms.
- s-type rooms: Always just 1 room (holds a max of 2 electrons).
- p-type suites: A set of 3 rooms (holds a max of 6 electrons).
- d-type suites: A set of 5 rooms (holds a max of 10 electrons).
- f-type suites: A set of 7 rooms (holds a max of 14 electrons).
- Checking in the 47 Electrons of a Silver Atom
- The electrons fill the “rooms” starting from the basement (closest to the nucleus) and moving up, never putting a third tenant in a two-person room.
- Floor 1: 1s room → 2 electrons
- Floor 2: 2s room (2) + 2p suite (6) → 8 electrons
- Floor 3: 3s room (2) + 3p suite (6) + 3d suite (10) → 18 electrons
- Floor 4: 4s room (2) + 4p suite (6) + 4d suite (10) → 18 electrons
- Floor 5: 5s room → 1 lone electron
- Total: 2 + 8 + 18 + 18 + 1 = 47 electrons.
The Pauli principle is a form of “quantum pressure.” Once the 1s room has two electrons, it’s “Full.” A third electron is mathematically forbidden from entering. It is forced to go to the next available room on a higher floor.
Notice that the first 46 electrons are all neatly paired up. But the 47th electron is a “lone ranger” all by itself in the 5s room on the top floor. It has no partner to cancel its magnetic spin.
This one tiny, unpaired electron makes the entire Silver atom chemically reactive. It has an ‘open slot’ and is desperate to bond with another atom to find a partner for its lone electron.
Part 2: The Shape of the Wave – What a “Room” Really Is
So, why does a p-suite hold exactly 6 electrons? To answer this, we have to stop thinking of electrons as tiny balls and start thinking of them as what they are: standing waves.
Imagine tapping a balloon. The entire surface vibrates, but it doesn’t travel anywhere. An electron in an orbital is like that—a 3D vibration. The “cloud” we see in diagrams is simply the area where this wave is “ringing” the loudest.
Because we live in a 3D world, a p-type wave can orient itself in three perpendicular directions:

- px orbital: Two lobes of the wave vibrating along the X-axis.
- py orbital: Two lobes vibrating along the Y-axis.
- pz orbital: Two lobes vibrating along the Z-axis.
These are the three “rooms” in the p-suite. According to the Pauli Exclusion Principle, each room can hold two electrons if they have opposite spins (one “Up,” one “Down”).
3 orbitals × 2 electrons each = 6 electrons.
A common mistake is to think one lobe holds one electron. This is incorrect. An electron in the px orbital exists in both lobes at the same time.
The lobes themselves have a mathematical property called phase, labeled “+” and “-“.
This doesn’t mean electric charge (the electron is always negative!). Think of it like a guitar string: when it vibrates, part of the string goes up (crest, “+”) and part goes down (trough, “-“). For a stable p-orbital wave, one lobe must be a “crest” and the other a “trough.” Exactly at the nucleus, between the lobes, the wave value is zero. This is a “node,” a dead zone where there is zero probability of finding the electron.
Part 3: The Cosmic Handshake – Rule 2: Waves Love to Harmonize
Now we get to the magic. When atoms get close, their electron waves overlap. This is where chemical bonds come from, and it’s all about how the wave phases interact.
1. Constructive Interference (The “Glue”)
If a plus-phase lobe from Atom A overlaps with a plus-phase lobe from Atom B, their waves add up. They “harmonize.” This creates a dense region of negative charge right between the two positive nuclei. This electron density acts like cosmic superglue, pulling both nuclei towards it and locking the atoms together. This is a stable Bonding Orbital.
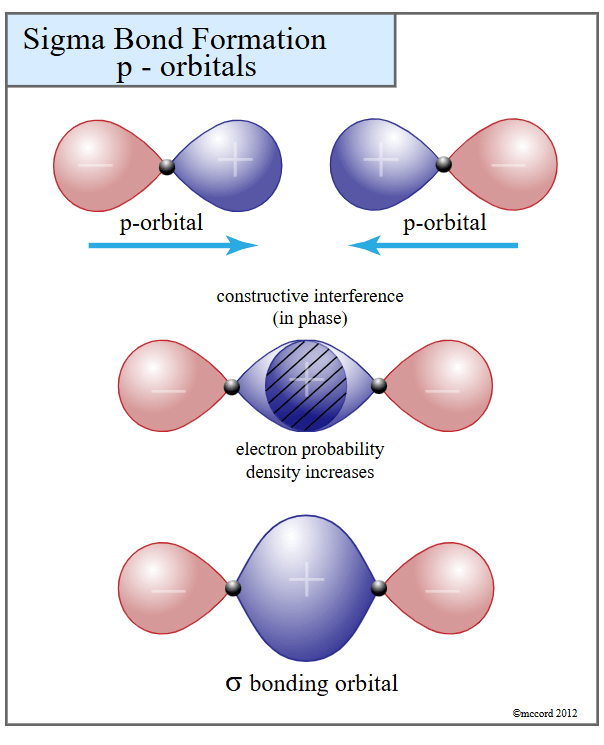
2. Destructive Interference (The “Wall”)
If a plus-phase lobe meets a minus-phase lobe, they are out of sync. They cancel each other out. The electron wave is pushed away from the center, leaving a “dead zone” (a node) between the nuclei. With no negative glue, the two positive nuclei “see” each other directly and repel violently. This is an Anti-bonding Orbital, and if electrons are forced into it, the molecule flies apart.
This is why molecules have specific shapes. In a water molecule (H₂O), the p-orbitals of the Oxygen atom stick out at roughly 90-degree angles. Two Hydrogen atoms “hook on” where their wave phases can match up constructively with Oxygen’s lobes. This perfect phase-matching is what forces the water molecule into its famous bent “V” shape.
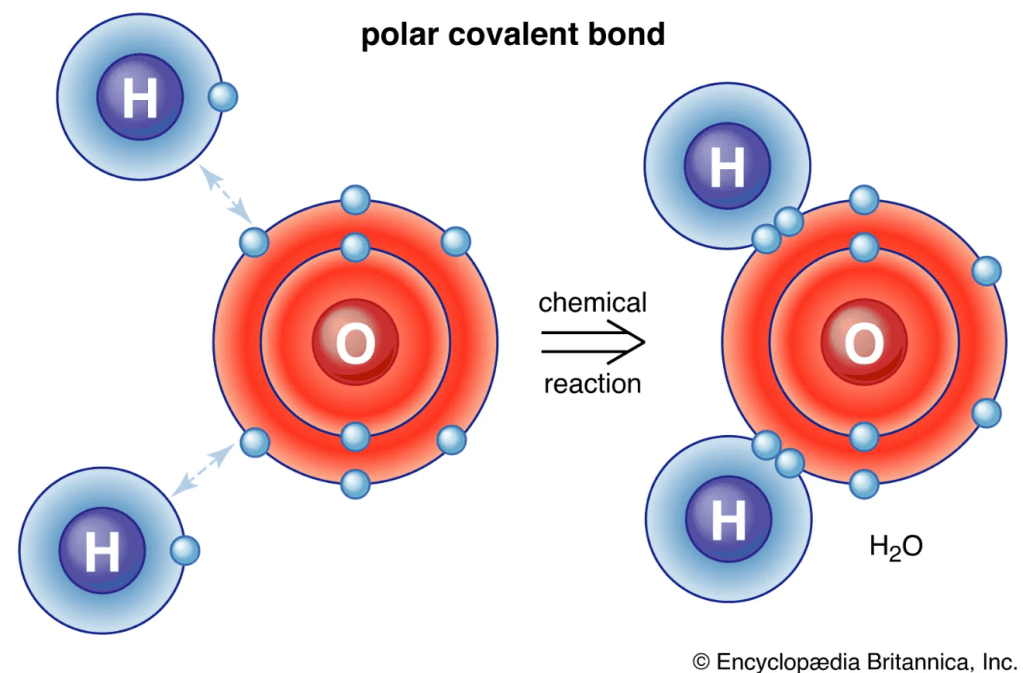
Part 4: The Rules in Action – Who Gets to Bond?
So, can any atom form a bond? No. An atom will only bond if it has an “opening”—an orbital with a single, unpaired electron.
To see how this works, we need one final detail: Hund’s Rule.
Hund’s Rule
This rule states that electrons are like people boarding a bus: they prefer to take their own empty seat (orbital) before they start pairing up with someone else.
Because electrons have the same charge, they stay as far apart as possible until they are forced to share a space.
When they do share a space, they must have opposite spins to balance each other out. If an electron is sitting alone in an orbital, it is “unpaired” and chemically reactive, looking for a partner with an opposite spin.
Case Studies
1. The Case of Nitrogen (Atomic Number 7)
Nitrogen has an odd number of electrons, which makes it very clear why it needs to bond:
- 1st Shell (1s): 2 electrons with opposite spins (Full).
- 2nd Shell (Valence): 5 electrons total.
- 2s orbital: 2 electrons with opposite spins (Full).
- 2p orbitals (2px, 2py, 2pz): According to Hund’s Rule, the remaining 3 electrons will each take their own orbital rather than pairing up.
- The Result: You have one electron in 2px, one in 2py, and one in 2pz. Since all three are “alone,” Nitrogen needs three more electrons with opposite spins to fill those gaps. This is why Nitrogen typically forms three bonds (like in Ammonia, NH3).
2. The Case of Oxygen (Atomic Number 8)
Even though Oxygen has an even number of electrons, Hund’s Rule still leaves it “unsatisfied”:
- 1st Shell (1s): 2 electrons with opposite spins (Full).
- 2nd Shell: 6 electrons total.
- 2s orbital: 2 electrons with opposite spins (Full).
- 2p orbitals: With 4 electrons left, the first 3 fill 2px, 2py, and 2pz individually. The 4th electron is then forced to pair up in the 2px orbital.
- The Result: This leaves one electron alone in 2py and one alone in 2pz. Because it has two unpaired electrons, Oxygen needs two more to reach stability.
3. The Case of Helium (2) and Neon (10)
These atoms follow the same rules but end up with no “lonely” electrons:
- Helium: Its only orbital (1s) is perfectly filled with 2 opposite-spin electrons.
- Neon: Every orbital (1s, 2s, 2px, 2py, 2pz) has a complete pair of opposite-spin electrons.
- The Result: Because there are no empty spots and no unpaired electrons, these atoms have no reason to bond with anyone else.
The Bottom Line: Bonding isn’t about odd or even total numbers; it’s about whether Hund’s Rule leaves any electrons sitting alone in an orbital. If they are alone, they will bond to find a partner.
4. The Curious Case of Carbon (Number 6) : The quirky Behavior
To understand Carbon (Atomic Number 6), we apply Hund’s Rule again, but with a special twist called hybridization that makes Carbon the most versatile element for life.
1. The Starting Point (Ground State)
If we just look at Carbon’s 6 electrons normally:
- 1s orbital: 2 electrons with opposite spins (Full).
- 2s orbital: 2 electrons with opposite spins (Full).
- 2p orbitals (2px, 2py, 2pz): Following Hund’s Rule, the remaining 2 electrons sit alone in 2px and 2py, leaving 2pz empty.
Strictly looking at this, you might think Carbon only needs two bonds to fill those two “lonely” spots. But we know Carbon almost always forms four bonds (like in Methane, CH4). Here is how:
2. The Carbon Twist (Excitation & Hybridization)
When Carbon is about to bond, it “promotes” one electron from the full 2s orbital into the empty 2pz orbital. This gives it four unpaired electrons:
- One in 2s
- One in 2px
- One in 2py
- One in 2pz
These four orbitals then blend together to become four identical “hybrid” orbitals.
3. The Result: Four “Lonely” Guests
Now, Carbon has four orbitals, and each one contains exactly one unpaired electron.
- The Result: Carbon now has four “unfilled rooms.” It is extremely “hungry” and searches for four more electrons with opposite spins to complete itself.
The Bottom Line: Because Carbon can spread its electrons out into four separate rooms, it can form four strong bonds. This ability to link up in so many directions is why Carbon can build complex structures like DNA, proteins, and diamonds.
Why Carbon “Promotes” Electrons (and why others don’t always)
Actually, other atoms can and do move electrons (this is called excitation), but Carbon is the “superstar” of this move because the energy cost is very low compared to the massive “payback” it gets from forming four bonds instead of two.
In an atom, the 2s and 2p orbitals are very close in energy. For Carbon, it only takes a tiny bit of energy to “bump” one electron from the 2s up into the empty 2pz seat.
- The Reward: By doing this, Carbon goes from having 2 unpaired electrons to 4 unpaired electrons.
- The Result: Forming four bonds releases a huge amount of energy, which more than pays back the small “cost” of moving that electron.
Why not others?
- Oxygen and Nitrogen already have electrons in all their 2p seats (2px, 2py, 2pz). There is no “empty seat” to move a 2s electron into without forcing it to pair up immediately, which costs too much energy.
- Neon has every single seat in the 2nd shell full. There is nowhere for an electron to go unless it jumps to a completely new, much higher shell (the 3rd shell), which is too difficult.
5. Silver Atoms (Atomic Number 47) and Hund’s Rule
Silver is a heavy metal, but the bonding logic remains the same. Let’s look at its outermost “seat”:
- Silver’s Outer Shell: Silver has one lone electron sitting by itself in its outermost orbital (5s1).
- Hund’s Rule & Stability: According to the principles we’ve discussed, this electron is “lonely” and wants a partner with an opposite spin to reach a lower, more stable energy state.
When two Silver atoms meet:
- The Same-Spin Conflict: If both Silver atoms have a “Spin Up” electron, they cannot bond. The Pauli Exclusion Principle says two electrons with the same spin cannot occupy the same space. They will repel each other like two magnets with the same poles facing.
- The Opposite-Spin Bond: If one is “Spin Up” and the other is “Spin Down,” they satisfy the requirement for a pair. They merge their electronic “waves,” creating a pool of negative charge between the two positive nuclei. This acts like “negative glue,” holding the two Silver atoms together.
The “Flip”: Atoms are so “hungry” for stability that if two “Spin Up” Silver atoms approach, one can actually flip its spin to become “Spin Down” just so they can pair up and bond. This state is much lower in energy and much more stable than staying apart.
The Bottom Line: Silver bonds because it has one “unfilled room.” By finding a partner with an opposite spin, it completes that orbital and stays “satisfied.”
The Big Picture
All of chemistry, from the glint of silver to the coil of your DNA, is a grand performance governed by two beautifully simple rules—one of separation, the other of unity.
1. The Rule of Individuality (The Pauli Exclusion Principle): Every electron demands its own unique state, its own “address” in the atomic hotel. This quantum pressure is what creates structure. It forces electrons into shells and subshells, building the entire periodic table and giving each element its unique identity, like assigning different instruments to their sections in an orchestra.
2. The Rule of Harmony (Wave Interference): Electron waves seek to overlap in phase, like musical notes blending in harmony. This constructive interference is the “glue” of all chemical bonds, pulling atoms together into the specific shapes and molecules that form our world. Destructive interference, like clashing notes, pushes them apart.
These two principles—one of exclusion and one of attraction—are the conductor’s baton and the musical score for the grand, intricate symphony of matter. They are the fundamental logic of the universe itself.
Leave a comment