- Meet the Players: A Tale of Two Particles
- Key Differences: Photon vs. Electron
- Where Does Light Come From? The Birth of a Photon
- Visualizing the Unseen: The Electron’s Wavy Nature
- The Double-Slit Showdown: Same Picture, Different Story
- How They Are the SAME (The Visual Pattern)
- How They Are DIFFERENT (The Physical Nature)
- Practical Differences in Their Patterns
- The Bridge Between Matter and Light
- Anatomy of a Quantum Interaction: How Electrons Create Light
- The Electron “Cloud” (Sigma-x)
- The Birth of a Photon (Gamma)
- The Cherenkov Cone
- The Detector and “Measurement”
- Near Field vs. Far Field: The Handoff Zone
- Conclusion
- The Physics of Creation: How Energy Becomes Frequency
- The Electron’s “Internal” Frequency and its Mass
- The Birth of a Photon: The Energy “Kick”
- The Photonic Sonic Boom: The Cherenkov Cone
- A Tale of Two Wavelengths: The Practical Benefits
- Conclusion: A Flawless Handoff

At the heart of quantum mechanics lies a fascinating paradox that challenges our everyday intuition. Imagine taking two fundamentally different things—one a pure packet of energy, the other a tiny speck of matter—and discovering they can create the exact same visual pattern. This is the quantum riddle of the photon and the electron.
While both famously exhibit wave-particle duality, they are not the same. One is the very essence of light and energy, while the other is a fundamental building block of the universe we see and touch. Let’s dive into what makes them unique, how they are eerily similar, and how their deep connection sets the stage for the very creation of light itself.
Meet the Players: A Tale of Two Particles
Before we see how they behave, let’s establish their identities. On one side, we have the photon, and on the other, the electron.
- The Photon: Think of a photon as a single “packet” or quantum of light. It is pure energy.
- The Electron: The electron is a subatomic particle, a tangible piece of matter.
They are opposites in almost every crucial way. A photon has no mass and no electric charge, which allows it to travel at the ultimate cosmic speed limit—the speed of light (c). An electron, however, has a tiny but definite mass and a negative charge. This mass means it can move incredibly fast but can never actually reach the speed of light.
Their roles in the universe are also distinct. The photon is a force carrier; it’s the messenger for the electromagnetic force. The electron is a constituent of matter; it forms the shells of atoms and is the workhorse behind electricity.
Key Differences: Photon vs. Electron
| Feature | Photon | Electron |
|---|---|---|
| Identity | A “packet” or quantum of light/energy. | A subatomic particle of matter. |
| Mass | Massless (zero rest mass). | Has mass (though very tiny). |
| Charge | Neutral (no electric charge). | Negative charge ( |
| Speed | Always travels at the speed of light ( | Can move fast, but never reaches |
| Role | Carries the electromagnetic force. | Forms atoms and carries electricity. |
Where Does Light Come From? The Birth of a Photon
If photons are packets of energy, where do they originate? The answer almost always involves their counterpart: the electron. A photon is created whenever a charged particle experiences a change in energy. It’s the universe’s way of carrying away the excess energy from a disturbance. Here are the four primary ways this happens:
- Atomic Transitions (Electron Jumps): This is the most common source of the light we see. When an electron in an atom is excited to a high-energy orbit and then “falls” back to a lower-energy orbit closer to the nucleus, it must shed that extra energy. It does so by emitting a single photon.
- Acceleration of Charges: Whenever you shake, speed up, or sharply turn a charged particle like an electron, you are accelerating it. This acceleration creates ripples in the surrounding electromagnetic field, and these ripples are photons. This is precisely how radio antennas broadcast signals—by wiggling electrons back and forth.
- Thermal Radiation (Heat): Why does a hot stove glow red? As an object heats up, its atoms vibrate more and more violently. This constant jiggling causes their electrons to accelerate and de-accelerate, leading them to emit a continuous stream of photons. We perceive this stream as heat and, at high enough temperatures, visible light.
- Matter-Antimatter Annihilation: In the most extreme case, when an electron collides with its antimatter twin, the positron, they annihilate each other completely. Their entire mass is converted directly into pure energy in the form of very high-energy photons known as gamma rays.
Visualizing the Unseen: The Electron’s Wavy Nature
We know photons are waves of light, but how do we visualize an electron—a particle of matter—as a wave? Because it has mass, its wave isn’t a simple squiggly line. Instead, it’s best understood as a wave of probability.
- Diffraction Patterns: If you fire a beam of electrons through a thin crystal, they don’t just pass straight through. They spread out and form a distinct pattern of concentric rings. This diffraction is a classic “fingerprint” of wave behavior.
- The Double-Slit Buildup: In the famous double-slit experiment, if you shoot electrons one by one, each one appears as a single dot on the detector screen. But over time, these individual, seemingly random dots build up to form a classic wave interference pattern. The wave, therefore, describes the probability of where any single electron is likely to land.
- Atomic Orbitals: Inside an atom, we no longer imagine electrons as tiny planets orbiting a sun. Instead, quantum mechanics describes them as 3D “standing waves”—fuzzy clouds of probability called orbitals that surround the nucleus.
The Double-Slit Showdown: Same Picture, Different Story
This brings us to the core of the riddle. If you perform the double-slit experiment with photons and then with electrons, the resulting patterns can look identical. If you saw a picture of the results without labels, you often couldn’t tell which particle created it.
1. How They Are the SAME (The Visual Pattern)
Both particles, when treated as waves, create an interference pattern—a series of alternating bright and dark stripes.
- Constructive Interference: Where the peaks of the two waves align and add up, you get a bright stripe (for light) or a region with many electron hits.
- Destructive Interference: Where the peak of one wave meets the trough of another, they cancel out, leaving a dark stripe (for light) or a region where no electrons land.
Mathematically, the rule is the same for both: if a photon and an electron have the same momentum, they will have the same wavelength and thus produce an identical interference pattern.
2. How They Are DIFFERENT (The Physical Nature)
While the final pattern is the same, what is actually “waving” is completely different.
- For Photons, it’s an Electromagnetic Wave: A photon’s wave is a real, physical oscillation of electric and magnetic fields. These fields carry force and can travel through the vacuum of space forever at the speed of light.
- For Electrons, it’s a Probability Wave: An electron’s wave (described by its wave function, ψ) is not a physical field shaking. It is an abstract, mathematical wave of probability. The height of the wave at any point tells you the likelihood of finding the electron at that specific location.
| Feature | Photon Wave | Electron Wave |
|---|---|---|
| What’s waving? | Electric/Magnetic Fields | Probability Amplitude |
| Medium | None (Vacuum) | None (Quantum Field) |
| Interaction | Can pass through each other | Repel other electrons (Charge) |
| Scale | Patterns are usually millimeters wide | Patterns are often at the atomic scale |
3. Practical Differences in Their Patterns
These fundamental differences lead to practical distinctions in how their waves behave:
- Scale: Because electrons have mass, they typically have much, much shorter wavelengths than visible light for a given energy. This is the secret behind Electron Microscopes, which use the tiny wavelength of electrons to see objects thousands of times smaller than what a light microscope can resolve.
- Bending: You can easily steer a beam of electrons with a magnet because they are charged. This allows you to bend and focus their wave patterns. You cannot do this to photons with a magnet; you must use physical lenses, mirrors, or gravity.
- Orbitals: Electrons form unique, stable “standing wave” patterns around an atom’s nucleus—the orbitals. Photons do not “orbit” nuclei; they are either absorbed by an electron, emitted by one, or simply pass by.
The Bridge Between Matter and Light
We’ve established that photons and electrons are fundamentally different entities that can, under the right circumstances, produce the same quantum pattern. But the most profound part of their story isn’t their difference, but their intimate connection.
As we saw, photons are almost always born from the actions of electrons—jumping between energy levels, getting shaken around, or annihilating. An electron, a particle of matter, gives birth to a photon, a particle of light. This interaction is the engine that drives everything from the light of the sun to the Wi-Fi signal carrying these words to you.
But how does this happen? What is the step-by-step process that allows a tiny particle of matter to create a massless packet of pure energy? Now that we understand the players, it’s time to explore the main event.
Anatomy of a Quantum Interaction: How Electrons Create Light
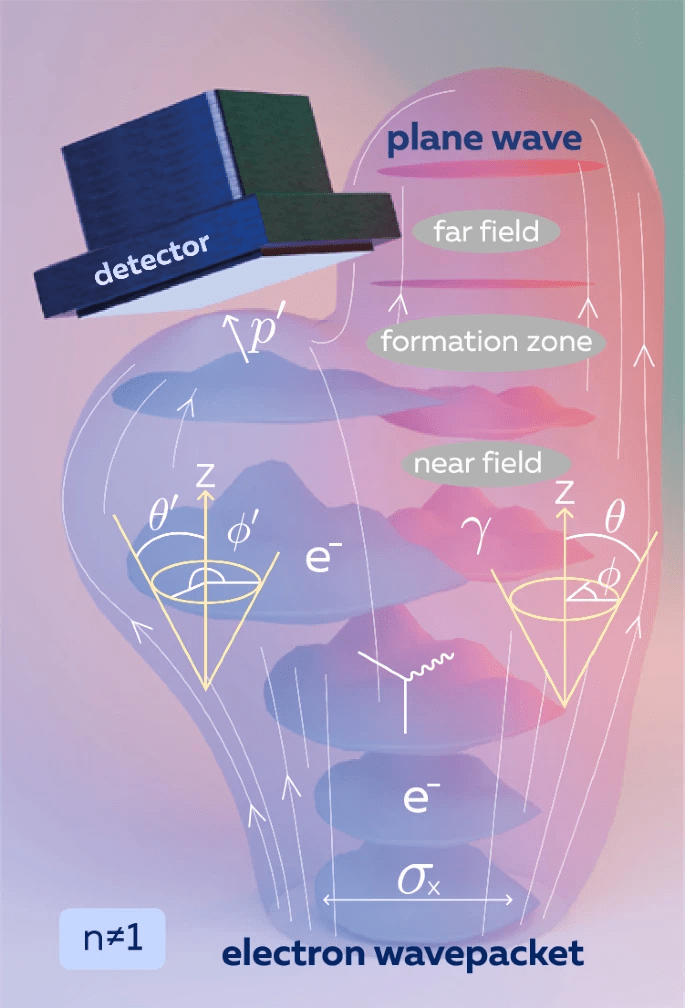
Description of Image: “Catching the Wave ->This visualization illustrates Quantum Electrodynamics (QED) in action. As an electron wavepacket (blue) travels through a medium, it disturbs the surrounding electromagnetic field, causing a photon (pink) to ‘peel away’ and become an independent wave. This represents the exact moment matter creates light.”
This diagram illustrates a high-level Quantum Electrodynamics (QED) interaction. It shows exactly how an electron “births” a photon as it moves through a material.
1. The Electron “Cloud” (Sigma-x)
The blue shapes labeled e- are the electron wavepackets. The symbol Sigma-x represents the initial “fuzziness” or size of that electron cloud. In the quantum world, the electron is not a single point; it is a localized wave moving upward through the medium.
2. The Birth of a Photon (Gamma)
The pink clouds labeled Gamma represent the photon field. In physics, an electron doesn’t just “contain” a photon; rather, as the electron moves, it disturbs the electromagnetic field around it.
- The Separation: In the “Formation Zone,” you can see the pink photon field literally peeling away from the blue electron.
- The Medium (Refractive Index n): This process happens because the electron is traveling through a material (like glass or water) where the Refractive Index (n) is not 1. In these materials, light travels slower than it does in a vacuum, allowing the electron to “outrun” its own field.
3. The Cherenkov Cone
If the electron moves faster than light can move in that specific medium, it creates a “sonic boom” of light. This is the Cherenkov Cone (the gold conical shapes).
- The Angle (Theta): The angle of this cone tells scientists exactly how fast the electron was moving.
- Near Field vs. Far Field: In the “Near Field,” the photon and electron are still “tangled” together. By the time they reach the “Far Field,” the photon has become a clean, flat Plane Wave (the pink disk at the top), completely independent of the electron.
4. The Detector and “Measurement”
The black box at the top is the Detector. This represents a fundamental rule of quantum mechanics:
- Wave Function Collapse: If we use the detector to measure the electron’s Momentum (p-prime), we “collapse” its wave function.
- The Observation Effect: The perfect Cherenkov cone only forms completely if the electron remains undetected while it is traveling. If we “peek” at the electron too early, we disrupt the delicate formation of the light wave.
Near Field vs. Far Field: The Handoff Zone
The distinction between the Near and Far Fields is crucial to understanding how a photon becomes independent.
In the Near Field, the electron and the photon are still “shaking hands.” They are physically overlapping and energetically tied together as a single, messy system. At this close range, the photon hasn’t fully formed into a clean wave yet; it is still part of the electron’s immediate “wake” as it moves.
In the Far Field, this bond finally breaks. The photon detaches completely and begins traveling as an independent, flat wave (a “plane wave”). Once they reach this stage, the “birth” is complete. The light travels away on its own, regardless of what happens to the electron that created it.
Conclusion
Once the photon escapes into the Far Field, it begins a life of its own, traveling at the speed of light until it hits another piece of matter. Crucially, the photon does not lose energy as it travels through a vacuum; its “color” or frequency stays exactly the same because there is no friction in empty space. It isn’t so much like a soul leaving a body, but more like a boat creating a wake in the water. The wake (the photon) carries away some of the boat’s energy, but once the wave is formed, it ripples across the lake independently of the boat that started it.
The “leftover” electron is now a low-energy version of its former self. Because it had to give up some of its kinetic energy to “pay” for the creation of the photon, the electron slows down. In the world of particle physics, this is a beautiful balancing act of conservation: the electron loses exactly as much energy as the photon carries away. This “tired” electron continues to move through the medium, but with less momentum, eventually settling into a lower-energy state or being captured by a nearby atom.
The Physics of Creation: How Energy Becomes Frequency
The diagram above gives us a stunning visual of an electron birthing a photon. But how does this process work on a fundamental level, and why do the electron and photon have different properties? Having witnessed their interaction, we now arrive at the heart of their relationship: the moment of creation.
This is the most “pleasantly surprising” part of the handoff: a photon is not actually a “piece” of an electron. Think of it like a tuning fork (the electron) hitting a pool of water (the electromagnetic field). The fork has its own vibration, but the ripple it creates in the water can have a completely different frequency depending on how hard the fork hit the surface.
Let’s break down why their frequencies diverge and how this process unfolds.
1. The Electron’s “Internal” Frequency and its Mass
The starting point is the electron itself. Because an electron has mass, it has an incredibly high “internal” frequency. It is a very “heavy,” fast-vibrating wave. This high frequency gives it a tiny wavelength, which, as we will see, is why it’s so beneficial for microscopy—it can “see” tiny details like atoms. This inherent, high-frequency ripple is the electron in its resting or moving state, ready to interact with the universe.
2. The Birth of a Photon: The Energy “Kick”
When the electron slows down or changes direction, it “kicks” the electromagnetic field. The frequency of the resulting photon depends entirely on how much energy the electron gave up during that specific kick. If the electron just “taps” the field, it creates a low-frequency radio wave; if it “slams” into the field, it creates a high-frequency X-ray.
To produce a photon of a specific frequency, you must give the electron an amount of energy exactly equal to E = hf, where E is energy, h is Planck’s constant, and f is frequency. In practical terms, this usually means accelerating the electron through a voltage such that its kinetic energy matches the desired photon’s energy.
| Photon Type | Frequency ( | Required Electron Energy (approx.) |
|---|---|---|
| Radio Wave | 100 MHz | 0.0000004 eV |
| Green Light | 545 THz | 2.25 eV |
| Ultraviolet | 10 PHz | 41.3 eV |
| X-Ray | 30 EHz | 124,000 eV (124 keV) |
| Gamma Ray | 300 EHz | 1,240,000 eV (1.24 MeV) |
As you can see in the reference table provided, the energy required to create different types of photons varies enormously, from the tiny fraction of an electron volt (eV) for a radio wave to over a million eV for a gamma ray.
To convert a specific frequency into the required electron energy, you use the Planck-Einstein relation.
- The Formula: E = hf
- E: Energy in Joules (J).
- h: Planck’s constant (6.626 x 10⁻³⁴ J·s).
- f: Frequency in Hertz (Hz).
To get the energy in electron volts (eV), you simply divide the result in Joules by 1.602 x 10⁻¹⁹ (the charge of a single electron).
3. The Photonic Sonic Boom: The Cherenkov Cone
One of the most mind-blowing illustrations of this energy transfer is the Cherenkov effect. The beautiful gold cone only forms when the electron travels faster than light in that medium. While nothing can outrun light in a vacuum (c), an electron can punch through a material like glass or water faster than light can. When it “breaks the light barrier,” it creates a photonic sonic boom. This is why nuclear reactor pools glow with a haunting blue light—you are literally seeing “tired” electrons shedding their excess energy as they outrun the light they are creating.
A Tale of Two Wavelengths: The Practical Benefits
This difference in their nature provides practical benefits. In the quantum world, an electron’s “wave ripple” has a much shorter wavelength than a photon of visible light. Because a wave can only resolve objects larger than its own wavelength, this is the secret to the Electron Microscope.
- The Electron (Near Field Probing): The electron’s tiny, high-frequency ripples act as a “near-field” probe. While visible light photons have long waves that blur out at the atomic level, the tight ripples of a fast-moving electron can “see” details thousands of times smaller, allowing us to map viruses and individual atoms.
- The Photon (Far Field Messenger): The photon, with its adjustable and often longer wavelength, is the perfect “far-field” messenger. It’s ideal for long-range communication and natural sight, carrying information over vast distances that a particle of matter cannot easily traverse.
Conclusion: A Flawless Handoff
When an electron drops to a lower energy state it imparts its excess energy into a photon rather than losing it. This “perfect accountant” of physics ensures that the energy surrendered by the electron is exactly equal to the energy carried away by the light, which then travels as an independent entity once it reaches the Far Field. As a self-sustaining wave, the photon moves through the vacuum of space without dissipating, maintaining its energy until it eventually hits another piece of matter and completes the transfer. This is the Conservation of Energy.
It is like a boat creating a wake in the water. The wake (the photon) carries away some of the boat’s energy, but once the wave is formed, it ripples across the lake independently. The “leftover” electron is now a low-energy, “tired” version of its former self, having given up some of its kinetic energy to “pay” for the creation of the photon. This “tired” electron continues on, having completed a flawless, high-speed handoff of energy that ensures nothing in the universe is ever truly lost.
Leave a comment