- The Story begins…………
- The Electron at Home: A Confined Standing Wave
- The Shape of Nothing: Why an Electron Is a Wave, and What That Actually Looks Like
- Part 1: The Trapped Wave
- Part 2: From Math Recipe to Physical Cloud
- Part 3: One Guitar String, Many Different Notes
- Part 4: Why Don’t All Electrons Have the Same “Shape”?
- Part 5: Electron ‘A’ vs. Electron ‘B’—A Deeper Look
- Case 1: Different “Frequencies,” Same Shape Family (e.g., 2p vs. 3p)
- Case 2: The Exact Same Orbital (e.g., Two Electrons in a 2pₓ Orbital)
- Conclusion: The Electron Isn’t the Wave, It’s the Water
- The Free Electron: A Traveling Wave Packet
- The Moving “Ripple”
- It Starts to “Spread Out”
- The Double-Slit Proof
- The “Pop”: When Does the Wave Stop?
- How We “Squeeze” the Wave: The Magic of Magnetic Lenses
- The principle behind the Electron Microscope
- Benefit of Electron Microscope
- Concept behind Particle Accelerator
- Conclusion: From a Tiny Ball to a Wave of Reality
The Story begins…………
At the heart of modern physics lies a concept so strange it famously “breaks everyone’s brain”: the dual nature of reality. Nowhere is this more apparent than with the humble electron.
- Is it a tiny ball of matter, or is it a wave?
- The short answer is: it’s both.
- More accurately, the electron is the wave.
This wave-like nature is governed by a fundamental rule of the universe: the Schrödinger Equation. While the name sounds intimidating, the idea is simple: it’s the wave equation for quantum particles.

This equation holds perfectly whether an electron is orbiting an atom or flying freely through a vacuum. What changes, dramatically, is the shape of that wave.
Let’s dive into the secret life of an electron, from its stable home to its journey into the wild.
The Electron at Home: A Confined Standing Wave
When an electron is “trapped” within an atom, it’s held in place by the pull of the positive nucleus. This confinement forces its wave into a specific, stable form known as a Standing Wave.
Think of a guitar string tied down at both ends. When you pluck it, it vibrates in a fixed pattern. It doesn’t travel up or down the neck; its energy is contained within a defined space. An electron in an atom is just like that.
The nucleus acts as a “leash,” preventing the electron’s wave from escaping.
This confined, vibrating wave of probability is what we visualize as an “electron cloud” or orbital.
The electron isn’t a little speck flying around inside this cloud; the cloud is the electron, its existence smeared out into a spherical, wave-like shape.
The Shape of Nothing: Why an Electron Is a Wave, and What That Actually Looks Like
We’ve all seen the textbook picture of an atom: a tiny solar system with little electron-planets orbiting a central nucleus. It’s simple, intuitive, and completely wrong.
Most of us have moved past that and accepted the modern upgrade: the “electron cloud.” But this idea creates an even stranger paradox. We’re told that this cloud is the electron, and that all electrons in the universe are fundamentally identical.
So, technically, why should you read this? Because if the cloud is the electron, and all electrons are the same, why do these clouds come in such bizarrely different shapes? Why is one a sphere (s-orbital), another a dumbbell (p-orbital), and others look like four-leaf clovers (d-orbital)? Shouldn’t every electron-cloud look the same?
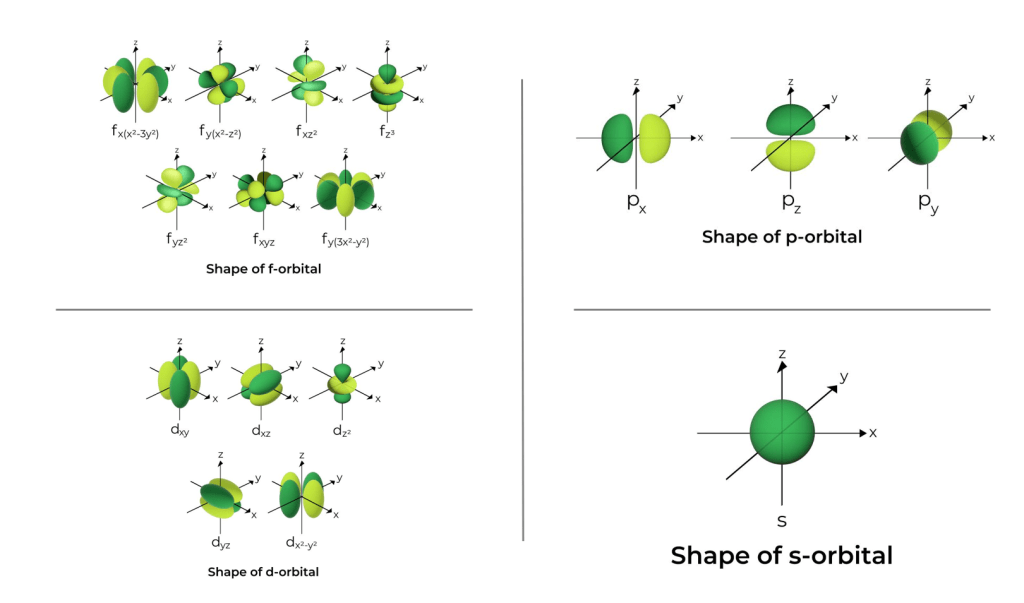
The answer takes us to the very heart of quantum mechanics, where an electron is more like a musical note than a tiny ball.
Part 1: The Trapped Wave
First, let’s establish what an electron in an atom really is. When an electron is “trapped” within an atom, it’s held in place by the powerful pull of the positive nucleus. The nucleus acts as a “leash,” preventing the electron’s wave from escaping.
This confinement forces its wave—yes, its wave—into a specific, stable form known as a Standing Wave.

Think of a guitar string tied down at both ends. When you pluck it, it vibrates in a fixed pattern. The wave’s energy is contained within a defined space; it doesn’t travel up or down the neck. An electron in an atom is just like that. This confined, vibrating wave of probability is what we visualize as an “electron cloud” or orbital.
Here is the most critical leap:
The electron isn’t a little speck flying around inside this cloud.
The cloud is the electron, its entire existence smeared out into a spherical, wave-like shape.
Part 2: From Math Recipe to Physical Cloud
This is where many of us get stuck. Moving from thinking of electrons as “little balls” to “waves of probability” is one of the hardest jumps in physics. How do we get these cloud shapes?
It all starts with a famous “recipe” known as the Schrödinger Equation.
- The Recipe (Schrödinger’s Equation): Think of this equation as a set of rules that governs how an electron’s wave must behave when trapped by a nucleus.
- The Mathematical Result (Wave Function, ψ): When you solve this equation for an atom, you get a purely mathematical description called a wave function (represented by the Greek letter ψ, or psi). This function contains all the information about the electron, but it isn’t something you can “see.” It can even have negative values, which doesn’t make physical sense for a probability.
- The Physical Picture (The Electron Cloud, ψ²): To get the physical “cloud,” you take that wave function and square it (ψ²). This gives you the probability density—the regions in space where the electron is most likely to be found. This is the cloud we visualize.
In short: The Schrödinger Equation is the math; the cloud is the physical interpretation of that math.
Part 3: One Guitar String, Many Different Notes
So if it’s all from one equation, why the different shapes?
The s, p, and d orbitals are not different from the wave equation; they are specific solutions to it.
Let’s go back to our guitar string. The “wave equation” for that string tells you how it can vibrate. Depending on how much energy you give it (how hard you pluck it), it can vibrate in different patterns:
- Low Energy: A simple, single arc (the fundamental note).
- High Energy: More complex patterns with multiple bumps (harmonics).

It’s the same for an electron:
- s orbitals: The simplest, lowest-energy “vibration” possible. This is the fundamental note, which results in a simple sphere.
- p and d orbitals: More energetic and complex “vibrations.” These are the harmonics, which create dumbbell or cloverleaf shapes.
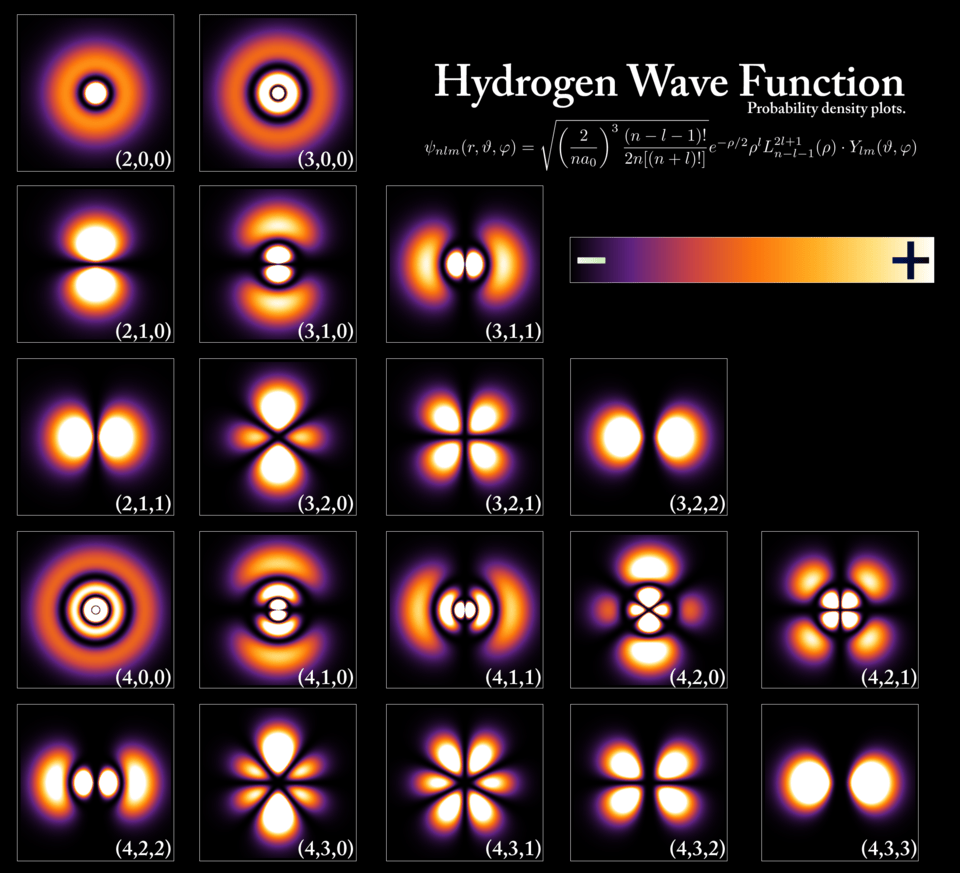
They are all results of the same Schrödinger equation, just with different energy and “twisting” (angular momentum) values plugged in.
Part 4: Why Don’t All Electrons Have the Same “Shape”?
This brings us back to our central question. If the orbital is the electron, shouldn’t they all look the same?
The “shape” of an electron isn’t a permanent physical feature like the color of your eyes; it’s a state of being based on its energy and environment. Two principles explain this diversity:
- Energy Levels: Just like a musical note’s wave pattern changes when you play a higher frequency, an electron’s wave shape changes when it has more energy. Higher energy allows for more complex, harmonic vibrations.
- The Exclusion Principle: In simple terms, electrons are “picky roommates.” The Pauli Exclusion Principle states that no two electrons can occupy the exact same quantum state in the same atom. To stay in the same atom, they have to differentiate themselves by taking on different energy levels or “shapes” (s, p, d, f).
So, while every electron is identical in mass and charge, their waves look different because they are vibrating at different frequencies and patterns to stay organized within the atom.
Part 5: Electron ‘A’ vs. Electron ‘B’—A Deeper Look
If every electron has a different energy, it feels like there should be an infinite number of shapes. So what is the real difference between the p cloud of electron ‘A’ versus electron ‘B’?
The reason we only see s, p, d, and f shapes is that these are “families” of vibrations, determined by a property called Angular Momentum (how the wave “twists” around the nucleus).
Let’s compare two electrons.
Case 1: Different “Frequencies,” Same Shape Family (e.g., 2p vs. 3p)
A 2p electron and a 3p electron both have the dumbbell shape because they have the same angular momentum. But the 3p electron has more energy (a higher frequency).
This changes two things:
- Size: The 3p orbital is much larger and extends further from the nucleus.
- Internal Structure: The 3p orbital has “ripples” inside it, called radial nodes. A node is a surface where the probability of finding the electron is exactly zero.
Think of it like this:
- A 2p orbital is like two solid balloons. The electron lives inside those balloons.
- A 3p orbital is like a small balloon placed inside a larger, hollow balloon. There is a “dead zone” (the node) between the inner part and the outer shell. The electron can be in the inner part or the outer shell, but never in the gap between them. This happens because the higher-energy wave has to “wiggle” more, creating zones of cancellation.
Case 2: The Exact Same Orbital (e.g., Two Electrons in a 2pₓ Orbital)
If two electrons are in the exact same orbital, they have the same shape, size, and frequency. To follow the Pauli Exclusion Principle, they must differ in one final way: Spin.
- Electron A has “Spin Up.”
- Electron B has “Spin Down.”
Think of two identical fans spinning in a room. They have the same shape and speed (frequency), but one is spinning clockwise and the other is spinning counter-clockwise.
Conclusion: The Electron Isn’t the Wave, It’s the Water
After all this, it’s better to refine our analogy one last time.
The electron is the “stuff” the wave is made of.
Just like “water” is a substance, but a “whirlpool” and a “calm surface” are different shapes that water can take depending on energy and conditions. Every electron in the universe is made of the same “electron-water,” but the nucleus and the energy level force that substance into a specific container—the orbital.
In short:
Electrons don’t have a shape; they inhabit a shape based on the rules of the atom they are allowed to occupy.
The Free Electron: A Traveling Wave Packet
So, what happens when we rip that electron away from its atom? The Schrödinger wave equation still applies, but the “shape” of the wave changes completely. The electron becomes a Wave Packet.
The Moving “Ripple”

Note: This is a Ripple Wave of moving electron. This Ripple is shown for simplicity
Instead of a stationary cloud, a free electron is like a single, moving ripple traveling through a pond. The wave equation now describes a “packet” of probability moving at a certain velocity. If you could see this wave, the center of that ripple is where you would be most likely to find the electron if you were to “click” a detector and measure it.
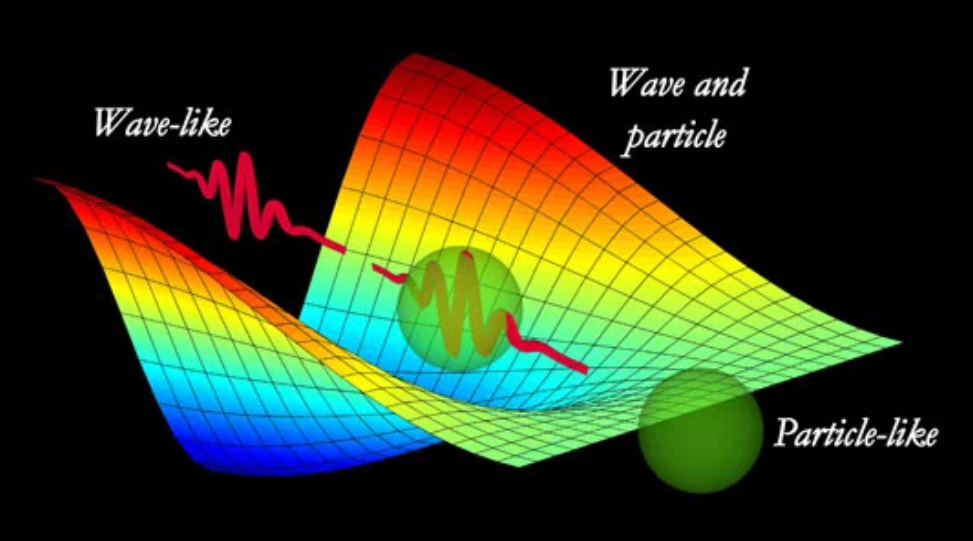
Note: Above is the image applicable to both Photon and Electron. It shows how can they behave both as wave and particle.
It Starts to “Spread Out”
Without the nucleus acting as a leash, a strange thing happens: the wave packet naturally spreads out over time. The longer a free electron travels without hitting anything, the “fuzzier” its location becomes. Its probability “cloud” literally gets bigger and thinner as it moves, meaning there’s a wider and wider area where it might be found.
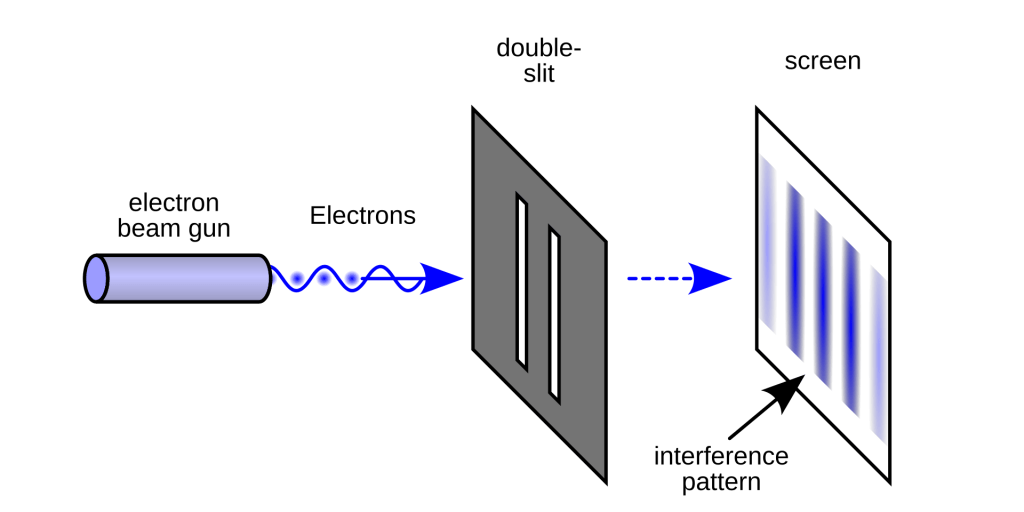
The Double-Slit Proof
How do we know this is true? The definitive proof comes from the famous Double-Slit Experiment. If you fire free electrons one by one at a barrier with two tiny slits, they don’t act like little bullets creating two distinct lines on the other side. Instead, they create an interference pattern—a series of many lines, just like light waves or water waves would. This proves that even when “flying” through a vacuum, the electron travels as a wave (governed by its wave function, ψ) that can interfere with itself.
The “Pop”: When Does the Wave Stop?
This leads to the most mind-bending part of quantum mechanics: Wave-Particle Duality.
The electron is always a wave. However, that wave has a very special property: whenever it interacts with something, it “collapses.” The wave equation only “stops” being a spread-out probability when the electron hits a fluorescent screen or a detector. At that exact moment, the entire wave “pops” into a single point, and we can say, “The electron is right here.”
- The “Pulse” View: When an electron is flying through a vacuum, it is a negative pulse (the wave packet). Its charge and mass are “smeared out” across that pulse. If you have a very wide wave packet, the “negative charge” is technically distributed across that whole area.
- The “Particle” View: The moment that pulse hits a target, the entire wave collapses. All the charge and mass that were spread out suddenly appear in one tiny spot. To our instruments, it looks like a particle just landed there.
The bottom line: The electron is always a wave.
The atom just gives that wave a specific, spherical shape.
Once free, the wave is just a traveling pulse that collapses into a point upon measurement.
How We “Squeeze” the Wave: The Magic of Magnetic Lenses
If free electrons are just fuzzy, spreading waves, how can we possibly use them to see things on a microscopic level? We can’t use glass lenses like in a camera, because the electrons would just get stuck in the glass. Instead, we “sculpt” these waves using Magnetic Lenses.
The principle behind the Electron Microscope
- The Problem: When electrons are boiled off a filament to create a beam, they spread out like a spray from a garden hose. Their “wave packets” are messy and wide.
- The Solution: We pass them through a donut-shaped electromagnet.
- The Physics: An electron is a moving charge, so it responds to magnetic fields. As the electron wave enters the magnetic field, the Lorentz Force pushes the charge, forcing the wave into a spiral motion toward the center.
- The Focus: By precisely tuning the strength of the magnet, we can “bend” the spread-out wave packet, forcing it to converge into an incredibly sharp point—a point thousands of times smaller than a human hair.
We take those “fuzzy” traveling waves, use magnets to focus them into a tiny “needle” of probability, and bounce them off a sample to see individual atoms. In essence, we use magnets to “sculpt” the electron’s probability cloud into the exact shape we need for our experiment.
Benefit of Electron Microscope
The primary benefit of this technology is unprecedented resolution. Because the wavelength of an electron is thousands of times smaller than that of visible light, focusing them into a tiny point allows us to “see” the nanoworld in high definition. This is the secret behind Electron Microscopes, which let scientists map the structure of viruses, design the microscopic transistors inside your smartphone’s CPU, and study the individual atoms in new materials. By using magnets to “squeeze” the electron spray into a sharp beam, we transform a messy cloud of matter into a precision tool that can capture images far beyond the physical limits of any glass lens.
Concept behind Particle Accelerator
This is the fundamental concept behind particle accelerators and proton guns. Whether you are using an electron, a proton, or a heavy ion, the principle remains the same: since these particles carry a charge, we can use magnetic and electric fields to steer, focus, and accelerate them. In a Proton Gun (used in cancer therapy), magnets focus a beam of protons to hit a tumor with pinpoint accuracy. In a Particle Accelerator like the Large Hadron Collider, these magnetic “lenses” are scaled up to massive proportions to keep particles on a precise track before smashing them together. The only real difference is the scale of energy and the specific particle being “pushed.”
Conclusion: From a Tiny Ball to a Wave of Reality
So, after our journey from the atom’s core to the freedom of a vacuum, what truly is the humble electron?
The answer requires letting go of the simple, intuitive picture of a tiny ball. Instead, the electron is best understood as a fundamental “stuff” of the universe, whose form is dictated entirely by its environment.
Let’s recap the complete picture:
1. The Electron at Home is a Standing Wave: When trapped inside an atom, the electron is not a planet orbiting a sun. It is the orbital cloud. Forced into confinement by the nucleus, its wave nature settles into a stable, vibrating pattern, much like a plucked guitar string. The different shapes—sphere (s), dumbbell (p), and cloverleaf (d)—are simply the different “notes” or harmonics it can play, determined by its energy. The cloud is the electron, its existence smeared out in a wave of probability.
2. The Free Electron is a Traveling Wave: Once ripped from the atom, the electron doesn’t become a bullet. It transforms into a traveling ripple—a “wave packet” of probability moving through space. Without the atom’s leash, this wave naturally spreads out, making its location increasingly uncertain the longer it travels.
3. The “Pop” of Duality: Here lies the heart of the mystery. The electron always travels as a wave. However, the moment it interacts with anything—a detector, a screen, another particle—its entire spread-out wave “collapses” into a single, definite point. It travels as a wave of potential but is measured as a particle of fact.
4. We are Masters of the Wave: This strange nature isn’t just a philosophical puzzle; it’s a tool. Using magnetic lenses in electron microscopes and particle accelerators, we have learned to “sculpt” these electron waves. We can take a fuzzy, spreading spray of probability and focus it into a needle-sharp point, allowing us to see and manipulate the world at the atomic scale.
Ultimately, the electron doesn’t have a dual nature; it is a quantum entity that reveals different faces depending on how we observe it.
It exists as a spread-out wave of potential until an interaction forces it to “choose” a single location.
The electron isn’t a particle hiding in a wave.
The wave is its true state, and the particle is simply the mark it leaves behind.
Leave a comment