- The Mystery
- Clue 1: The Mystery of the Two Lines (1922)
- The Detective’s Note: Why Silver Atoms?
- Clue 2: The Ghost in the Light (1924)
- The First Theory: An Impossible Spin (1925)
- The Final Twist: It’s Not Spin, It’s “Spin” (1928)
- The Verdict: It’s Just a Line of Code
The Mystery
- Science isn’t always a straight line from question to answer. Sometimes, it’s a detective story where the clues make no sense, the main suspect has an alibi, and the truth is stranger than anyone could have imagined. This is one of science’s greatest whodunits, and the mystery revolves around a single, unassuming particle: the electron.
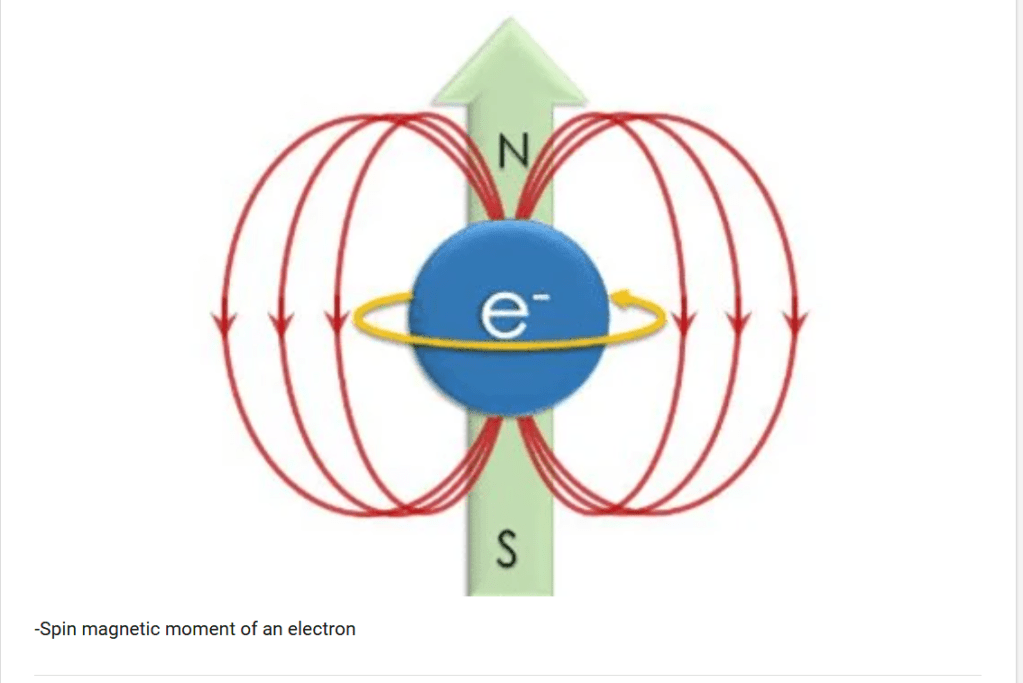
- For a time, physicists thought they had the electron figured out. It was a simple speck of negative charge and mass. But then, it started behaving strangely. It acted as if it had a secret identity—a powerful, intrinsic magnetism that defied all explanation.
- How could this be? An electron is a point particle, without a surface to “spin” like a planet. And even if it could, it would have to rotate faster than the speed of light to generate the magnetism observed.
- The alibi was solid; the simple explanation was impossible.
- Physicists were left with a ghost in the machine: a bizarre effect with no logical cause.
- They were forced to hunt for a culprit they couldn’t see, a fundamental property of the universe no one had ever dreamed of.
This is the story of how they cracked the case, not by observing a cause, but by working backward from its strange and undeniable effects. And our investigation begins with the first piece of evidence that blew the case wide open.
Clue 1: The Mystery of the Two Lines (1922)
The investigation begins with Otto Stern and Walther Gerlach. They wanted to test a simple idea: are the tiny magnets inside atoms oriented randomly, or are they “quantized”—stuck in specific directions?
To find out, they fired a beam of silver atoms through a cleverly designed magnetic field. If the atomic magnets were random, the beam should have smeared into a single, continuous line on their detector screen.
But that’s not what happened. The beam split into exactly two distinct paths.
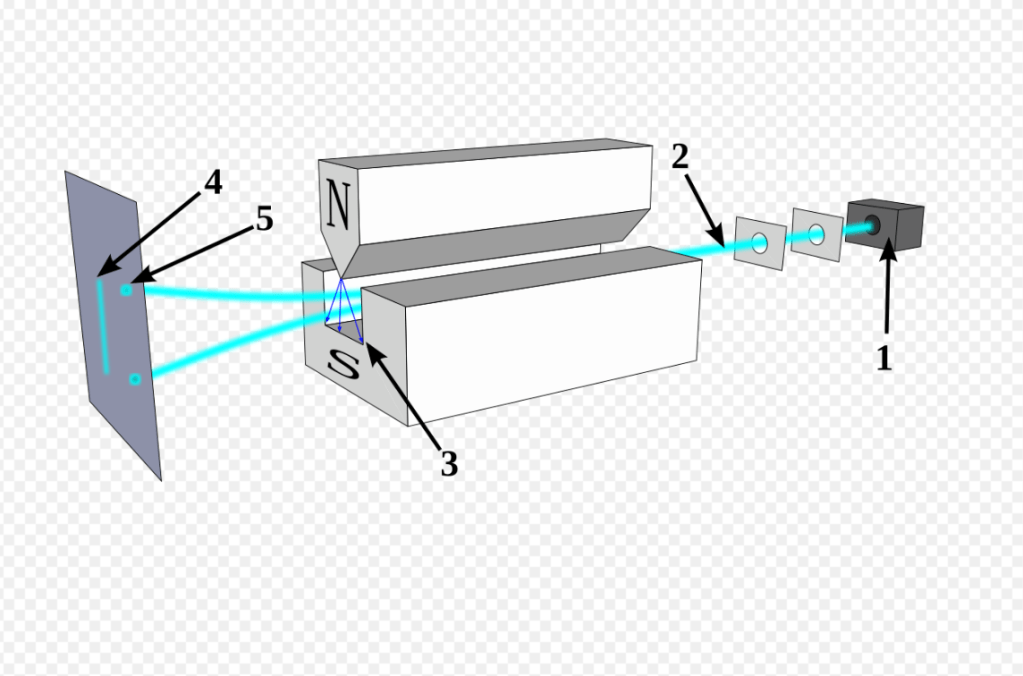

This result was baffling. Why only two? At the time, they guessed it was related to the electron’s “orbit,” but they had no idea they had just stumbled upon the first major clue.
The Detective’s Note: Why Silver Atoms?
Why not just use electrons? Because an electron’s charge would cause it to curve wildly in a magnetic field, obscuring the tiny effect they were looking for. A neutral silver atom was the perfect stealth vehicle. Its first 46 electrons are “paired up,” meaning their magnetic fields cancel each other out. This leaves a single, unpaired electron in the outer shell. The entire silver atom, therefore, acts like a transport box, carrying the magnetic signature of that one lone electron. The atom’s behavior was the electron’s behavior.
Clue 2: The Ghost in the Light (1924)
Meanwhile, other scientists studying the light given off by excited atoms noticed something equally strange. Certain spectral lines, like the famous yellow glow from sodium, weren’t single lines at all. Under high resolution, they were “doublets”—two lines incredibly close together.
It was the same “two-ness” mystery, just in a different form. The brilliant physicist Wolfgang Pauli realized that to explain this, the electron must have a fourth property—a fourth “quantum number”—that could only have two possible values. He called it a “classically non-describable two-valuedness.” He knew the clue was real, but he refused to give it a physical description.
The First Theory: An Impossible Spin (1925)
It took two daring graduate students, Samuel Goudsmit and George Uhlenbeck, to propose a name for this two-valued property: spin. They suggested the electron was literally spinning on its axis, like a tiny planet, creating its own magnetic field. This would perfectly explain the two paths in the Stern-Gerlach experiment (“spin up” and “spin down”).
It was a brilliant leap, but it immediately hit a wall.
The legendary physicist Hendrik Lorentz did the math. Given the electron’s known magnetism and its tiny size, he calculated how fast its surface would have to rotate. The answer was a disaster: the electron’s equator would be moving many times faster than the speed of light, breaking the most fundamental law of the universe.
The theory seemed dead on arrival. The electron had the properties of a spinning object, but it couldn’t possibly be a spinning object.
The Final Twist: It’s Not Spin, It’s “Spin” (1928)
The case was finally cracked not by observation, but by mathematics.
First, Llewellyn Thomas corrected the math using Einstein’s theory of relativity, fixing a key discrepancy and making the “spin” model fit the data from the spectral lines perfectly.
But the final proof came from Paul Dirac. In 1928, he formulated a new equation that combined quantum mechanics with special relativity. He wasn’t trying to solve the spin mystery; he was trying to create a more complete description of the electron. To everyone’s astonishment, the solution to his equation didn’t just allow for an electron with two magnetic states—it required it.
Spin wasn’t something you added to the electron. It emerged from the fabric of relativistic spacetime. It was a fundamental, built-in property.
Summary Table: Discovery Timeline
| Year | Discovery | Key Figure(s) | Impact |
|---|---|---|---|
| 1922 | Beam splitting in magnetic fields | Stern & Gerlach | First physical proof of “two-valued” magnetism. |
| 1924 | The “Fourth Quantum Number” | Wolfgang Pauli | Identified the need for an extra “slot” in electron code. |
| 1925 | The word “Spin” proposed | Goudsmit & Uhlenbeck | Linked the code to angular momentum. |
| 1928 | Relativistic Wave Equation | Paul Dirac | Proved spin is a fundamental, non-mechanical property. |
The Verdict: It’s Just a Line of Code
So, what is spin if it isn’t physical rotation?
- This is the core of the discovery.
- Scientists weren’t surprised that electrons had magnetism; they already expected that from the electron’s orbital motion. Instead, they observed a magnetic effect—the beam of silver atoms splitting into exactly two paths—that was completely unexpected and couldn’t be explained by existing theory.
- This forced them to conclude that the electron must possess an additional, intrinsic quantum property that was the true source of this magnetism.
- This previously unobserved property, which gives rise to the electron’s inherent magnetic moment, is what was ultimately named “spin”.
The modern understanding is that spin is an intrinsic property that is the fundamental source of the electron’s inherent magnetism. You could say that “spin” is the name we give to the quantum property that causes the magnetism of an electron we observe. The magnetic property is as fundamental to the electron as its charge, and spin is the engine that drives it.
An electron has two fundamental properties given to it by nature:
1. Negative Charge
2. Magnetic Property
- Think of it like a character in a video game that has a built-in “magnetism” attribute. The character isn’t carrying a physical magnet; this property is just a line of code in their fundamental definition.
- Similarly, an electron’s magnetism is like a built-in attribute in a video game’s source code.
- It is an intrinsic property called ‘spin.’
- While it behaves mathematically like angular momentum, the electron is not physically rotating on an axis—it has no volume to spin.
- Rather, spin is a fundamental ‘hard-coded’ value that exists without physical movement, naturally resulting in a magnetic moment
In this sense, spin is the mathematical label we’ve assigned to describe the electron’s physical magnetic polarity; a value of +1/2 simply denotes one specific North/South orientation, while -1/2 denotes the reverse. This is the key to understanding the experimental results:
- A “spin up” electron (+1/2) has its magnetic moment pointing down.
- A “spin down” electron (-1/2) has its magnetic moment pointing up.
Because a static electron doesn’t actually “spin” like a physical ball, this concept was reverse-engineered to account for the observed magnetism that couldn’t be explained by orbital motion alone.
We created the term “spin” to bridge the gap between our classical understanding of rotating charges and the reality of a stationary particle possessing intrinsic magnetic poles.
The entire structure of matter is built on a property that we only discovered by working backward from a mystery that, at first, seemed impossible.
Reference:-
Leave a comment